 Freshwater/Süßwasser
Freshwater/Süßwasser

Juwel Trigon 190

Juwel Rio


 Marine/ Meerwasser
Marine/ Meerwasser

Jaubert System
In 2000 as 63l "experiment".
- Plenum made by glueing 1cm rings of U-pvc tube under a plastic plate with holes in. Coarse filter wool on top prevented broken coral and the coral sand from dropping through.
- Needing more light, 2 x 14W tubes and in-hood controller, the water reached over 30°C. An external controller gave less heat.
- Heating: None needed.
- No filtration- A small filter, allowed wider surface flow than a recirculating pump
- Later I added a small hang-on box filter, the smallest Sander skimmer fitted nicely inside.

- Nov. 2010 changed to 96L aquarium- using Eheim undergravel filter plates, coarse coral then fine coral sand on top.
- More recent ideas suggest the 2 layers are unnecessary. Some succesfully use a deep sand bed with no plenum.
- Tunze Comline filter added (mainly for surface cleansing). Low wattage heater added.
- Cover with 2 x 24W T5. Changed later to 2 x 7W T5 LEDs (Blue and White).
Gobies, shrimps, hermit crabs, stony and soft corals.

'Berlin' System
- 130cm x 60cm x 60cm panorama with filter sump
- 2x turbelle low voltage with surge controller
- Deltec skimmer
- Giesemann 2x 250W 10,000 Kelvin HQI (8h daily)/ 2 x 39w actinic (12h daily)
- Reverse osmosis, top-up (3litres per day) using osmolator.
- UV lamp
- Tropic Marin salt and Biocalcium, Trace Elements self- prepared KI and SrCl2 solutions along with additional vitamins
Coral fish, shrimps, hermit crabs, stony and soft corals.



 Nano
Nano

 Slightly larger 'Scapers' Tank
Slightly larger 'Scapers' Tank
 'Scapers' tank setup.
'Scapers' tank setup.
 Chromaphyosemion bitaeniatum.
Chromaphyosemion bitaeniatum.
Water Chemistry
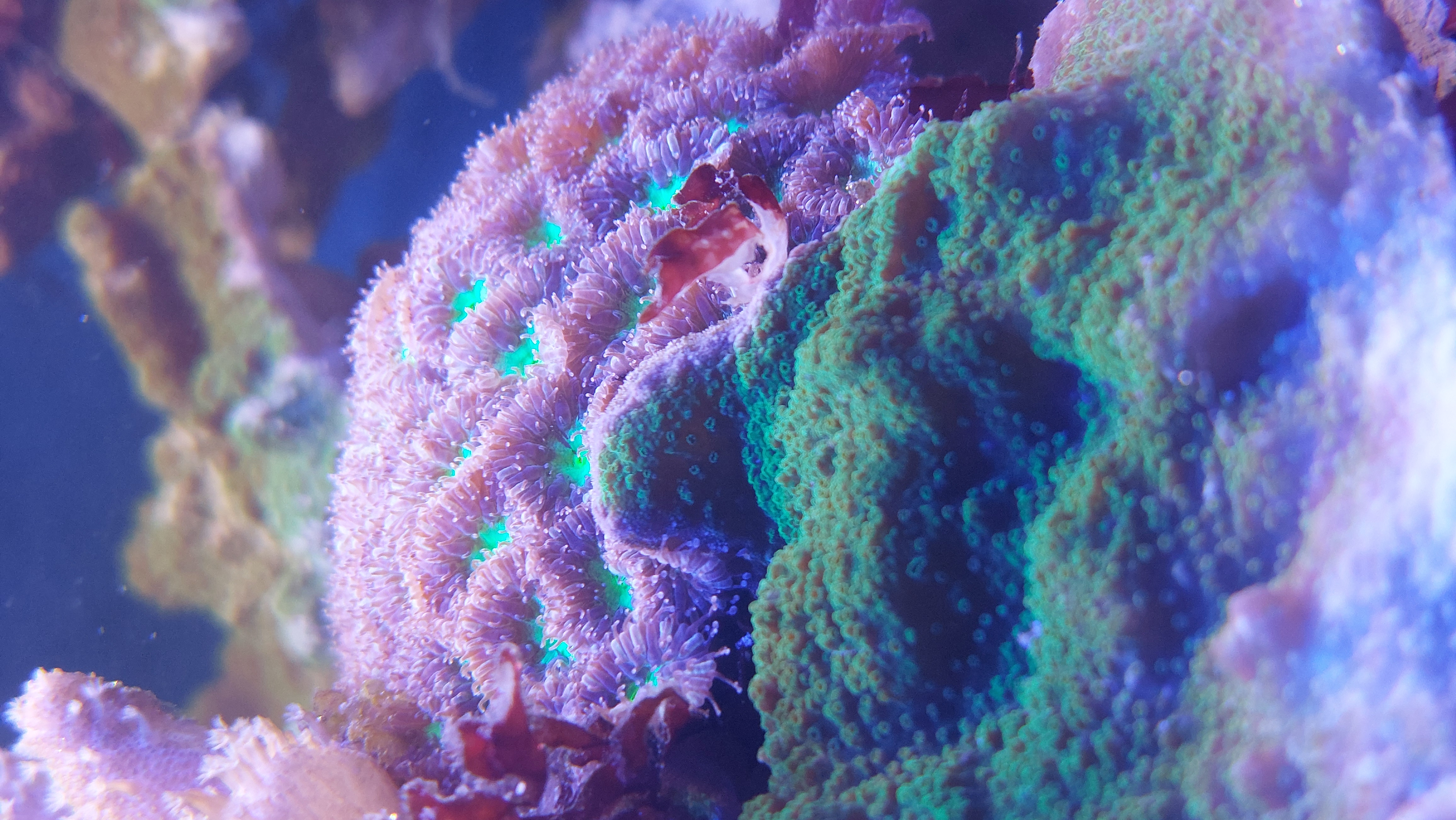
pH
Many freshwater fish come from regions with pH between 6 and 7.5, and will tolerate living in values slightly outside this. Many livebearers find slightly alkaline water ideal, and although tetras and rasboras will live in this water, breeding may involve conditions more akin to where they are from. Chocolate gouramis and parosphromenus species need acidic water pH 5-6. Discus prefer around 6. Rift lakes cichlids on the other hand like it alkaline around 8, and so my water is ok for them, at least as far as pH is concerned. Seawater has a pH of around 8.
Some fish have been bred commercially in ponds for generations, and may well have adjusted to different values.
What affects pH in an aquarium?
Wood can reduce pH, CO2 addition will reduce pH as may peat extracts and 'black water' tonics. Some stones are likely to raise it, those found in aquarium shops shouldn't to any degree.
Intense light, including afternoon sunshine, on an aquarium may cause an increase in photosynthesis, levels of carbon dioxide/ hydrogen carbonate may decrease, and the pH may rise significantly.
KH
this is 'carbonate hardness' but can also be seen as buffer capacity, since it binds with acidity, stabilising the water's pH. This one reacts with acids stopping a rapid drop in pH. It used to also be called temporary hardness, since it can be removed by boiling. This is the scale deposited in boilers and kettles.
Ca(HCO3)2 --boiling --> CaCO3 + H2O + CO2
GH
standing for 'general' hardness or 'GesamtHärte', this is the total [Ca+] and [Mg+] in water. Apart from these Calcium and Magnesium ions, there are one or two others present in such small amounts that we can safely ignore them. The part of the total hardness includes that which cannot be removed by boiling, and has been called 'permanent' hardness.
Conductivity
Mentioned in older books but not considered for the home aquarist because of the cost of instruments. Nowadays a small digital meter will cost below 30€. Water is a good solvent. On its way to us, it dissolves small amounts of minerals and gases. Some rock formations such as chalk will contain calcium and magnesium, whereas others such as granite and lava will not. The dissolved substances are present as and ions. These electrically charged particles will move in water when a voltage is applied, how much current flows corresponds to the quantity of minerals dissolved. BUT this means all minerals, not just those causing hardness.
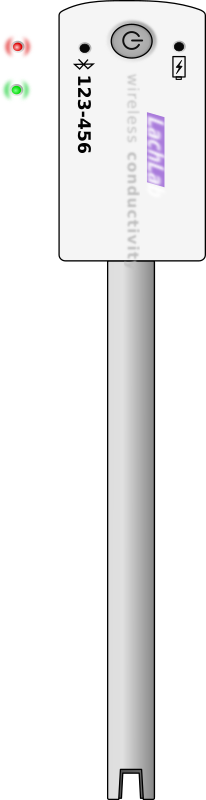
Aquarium water starts off with the same conductivity as tap water. Between 600 and 1000 µS in a hard, mineral rich water like mine, this is really quite hard as well. Reverse osmosis water from an aquarium setup has a conductivity of perhaps 10µS. Fish waste, fish food, water conditioners, fertilisers and other additives can change these values. Other things being equal, a significant rise would indicate it's time for your regular water change (max 30%).
The Nitrogen Cycle and the Aquarium
Nitrogen is essential to life. Plants take it up, helped by bacteria, animals eat plants, animals die and decompose, and bacteria free nitrogen again. This is 100 times simplified. What happens in aquariums, being at least a partially closed system, is an incomplete version of this.
Tests
- Combi test strips- the least accurate but the most convenient for a quick overview. Stick to well known makes and you'll get 6 or 7 parameters measured all at once. Good value.
- Liquid tests- can be selected according to the pH range required. Thes are made of combinations of special dyes called 'indicators' which change colour at particular pH values. Other tests rely on a colour change due to a chemical reaction. Some of the reagents may be caustic or be harmful in other ways. Read the instructions from beginning to end before use.
- pH meters- very good but only as accurate as their maintenance and calibration, electrodes do not last for ever. Conductivity meters, very accurate if kept clean.